PRODUCTS
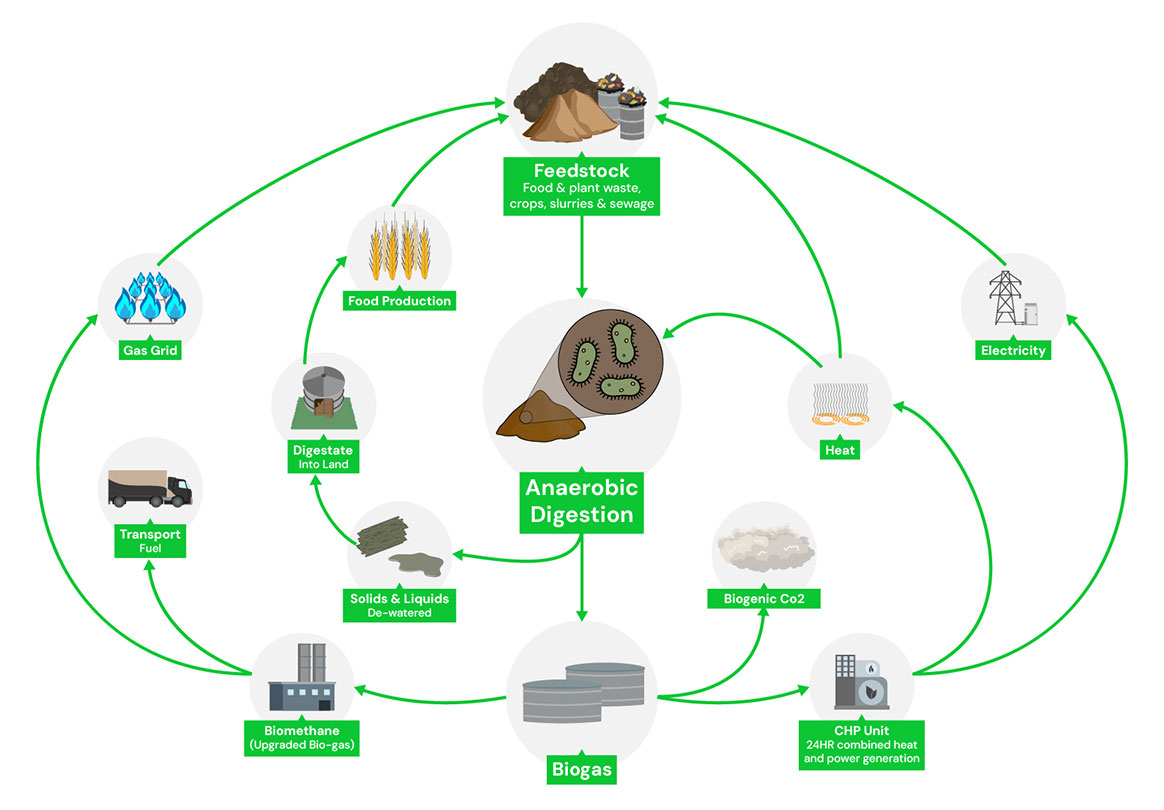
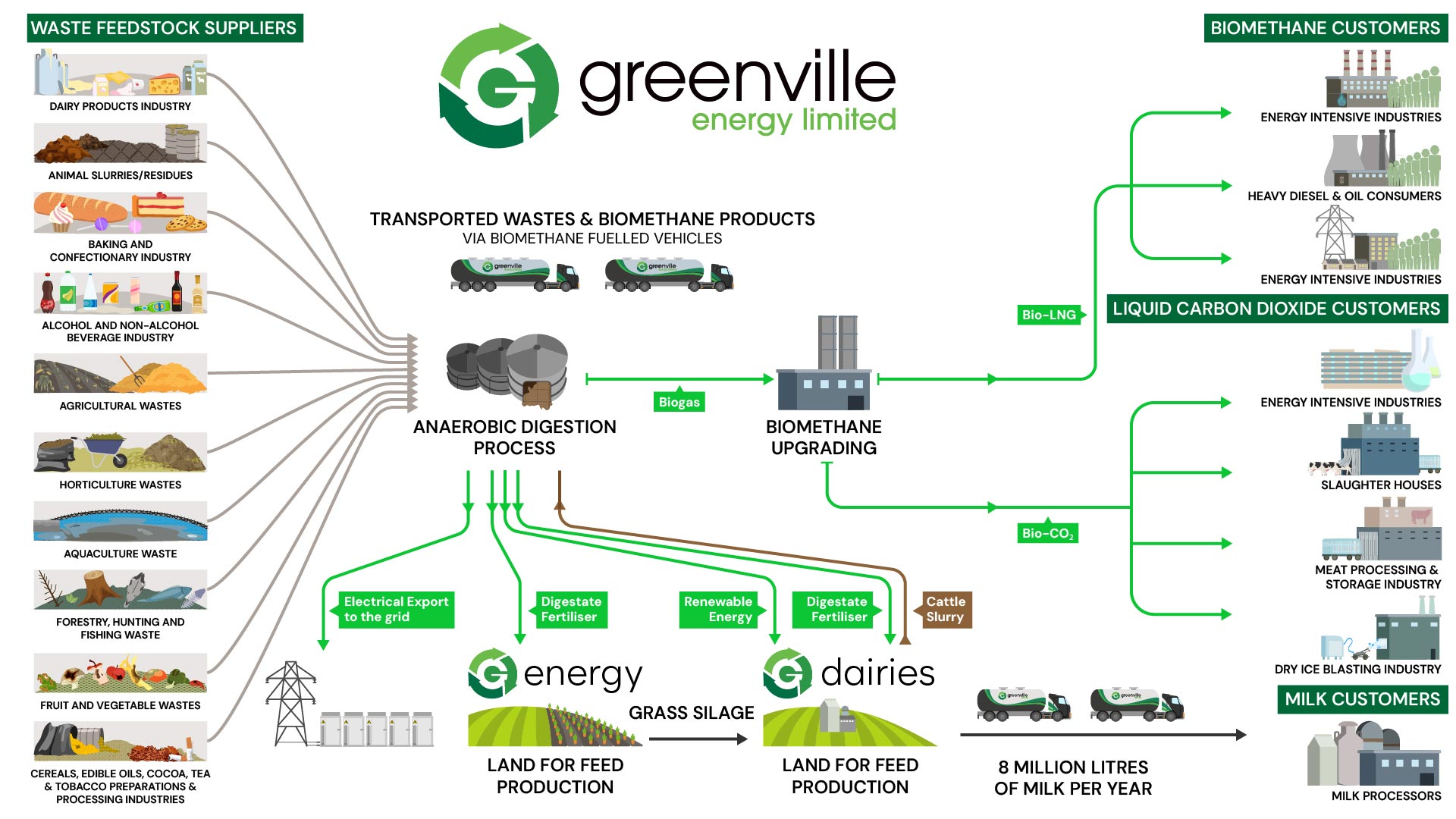
Biogas
At Greenville Energy, biogas production is an integral part of our commitment to renewable energy, waste management, and environmental stewardship.
Biogas, primarily composed of methane (CH₄) and carbon dioxide (CO₂), is generated through anaerobic digestion.
This process harnesses the natural breakdown of organic material—specifically waste materials like food waste and by-products from commercial food production—under oxygen-free conditions.
Importantly, Greenville Energy does not grow crops for energy production; all biogas is derived from waste resources, aligning with our sustainable and zero-emission objectives.

The Composition and Chemistry of Biogas
Biogas consists mainly of two primary gases: methane (CH₄) and carbon dioxide (CO₂), with trace amounts of other gases. Methane, a hydrocarbon gas, serves as the key energy carrier in biogas, providing the fuel for heat, power, and further refinement into biomethane. Here’s a detailed look at these molecules:


The Environmental Benefits of CH₄-based Biogas at Greenville Energy
Greenville Energy’s production process results in a methane-rich biogas that can be combusted cleanly with minimal air pollutants. By using waste-derived biogas as a renewable energy source, we significantly reduce reliance on fossil fuels and decrease greenhouse gas emissions. Importantly, our methane production is carbon-neutral because the CO₂ emitted during combustion is part of the natural carbon cycle, derived from renewable organic waste.
Through this approach, Greenville Energy not only fulfills current energy needs but also contributes to a sustainable energy future that aligns with stringent environmental standards and regulatory requirements. By producing clean, renewable energy from waste, we close the loop in energy production and contribute to a circular economy that values waste as a resource.